Application and Technology News

The Health Quarters: Esco Healthcare Quarterly Newsletter (Issue 1, May 2021)
Esco Healthcare unveils the first issue of its quarterly newsletter, The Health Quarters (The HQ), featuring its esteemed project highlight, informative behind-the-product technological principles and know-hows, as well as its latest innovative product advancements.

Esco Laminar Flow Trolleys: Aseptic Transport of Sterile / Aseptic Materials
Esco Laminar Flow Horizontal Trolley/ Laminar Flow Vertical Trolley (LFHT/LFVT) are mobile transport carts or trolleys providing ISO Class 5 clean air (as per ISO 14644-1). These laminar flow trolleys utilize a unidirectional airflow to eliminate contamination during the transfer of sterile/aseptic materials.

Integrable and Dependable: Esco Airstream® Fan Filter Units
Esco Airstream® Fan Filter Unit is a leading-edge fan filter unit (FFU) designed to provide HEPA/ULPA-filtered laminar airflow over a specific area. The FFU draws in ambient air from the top of the module, and exhausts clean filtered air vertically in a unidirectional (laminar) air stream toward its base.

Robust and Modular; Esco GB ships VBE to a Major Company in Ireland
Esco GB Ltd recently customized a 4ft Ventilated Barrier Enclosure (VBE) unit to suit the process needs of a major international company in Ireland.
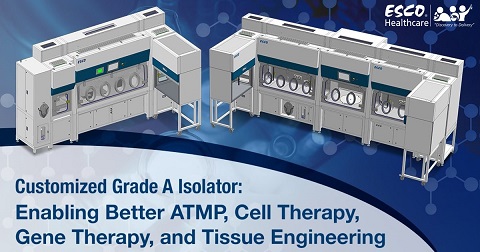
Cell Therapy Grade A Isolator: Enabling Better Biotherapeutics Manufacturing
A Swiss biologics contract development and manufacturing organization (CDMO) chose Esco Isoclean® Healthcare Platform Isolator – Inflatable Seal Model (HPI-IS) as the unit to be customized to meet it’s process requirements.

Esco launches new Streamline® Compounding Isolator in the Global Market
In line with the revised USP and Pharmaceutical Inspection Co-operation Scheme (PIC/S) guidelines, Esco opens the decade with its upgraded Streamline® Compounding Isolator (SCI) to continuously promote optimum patient health.

Integration Made Easy with Esco’s Robotic Filling Line Isolator
Esco’s highly flexible filling line isolators are the prime solution for the aseptic production of novel pharmaceuticals as they are customizable to suit the individual clients’ needs; this project proves just that.

Esco GB Ltd Engineers a Robotic Filling Line Isolator for a Major Pharmaceutical Company in Ireland
Esco’s highly flexible filling line isolators are the prime solution for the aseptic production of novel pharmaceuticals as they are customizable to suit the individual clients’ needs; this project proves just that.





