Why isolators are still preferred when handling HPAPIs?

Inherent risks associated with HPAPIs
Highly-potent active pharmaceutical ingredients (HPAPIs) show effects at much smaller doses. Unintentional exposure to these agents could potentially result in undesirable effects on operators.
Handling challenges for HPAPIs
Handling HPAPIs is different from other active pharmaceutical ingredients. Specialized containment is required to protect workers, facilities, and environment from HPAPIs. Moreover, as applied to other pharmaceuticals, product protection from contamination and losses is also required.



Type of Containment Options
| Band Category | OEB 1 | OEB 2 | OEB 3 | OEB 4 | OEB 5 | |||||||
| OEL (μg/m3) | 10,000 to 1,000 | 1,000 to 100 | 100 to 50 | 50 to 10 | 10 to 1 | 1 to 0.1 | 0.1 to 0.01 | |||||
| Equipment / Containment Systems | Local Exhaust Ventilation | |||||||||||
| Crossflow booth | ||||||||||||
| Downflow Booths | ||||||||||||
| Cone Valves | ||||||||||||
| Split Butterfly Valves (SBV) | ||||||||||||
| SBV with Air Extraction | ||||||||||||
| SBV with Washing | ||||||||||||
| Rapid Transfer Ports (RTP) | ||||||||||||
| Isolators | ||||||||||||
Source:
- Engineering Controls: Balancing Cost and Containment Hazard by P. Richards
- Occupational Exposure Banding by A. George
- Potent Product Process and Facility Design by S.C. Singhai
Why choose isolator then?
Benefits of Isolators
- Minimized exposure to operator
- Lowest cross contamination
- Smaller contact surface area
- Minimal energy requirements
- Material handling without exposure
- Ideal for aseptic processing
- Localized inert gas environment is possible
- Suitable for OEB 4 and 5 potent materials
Contamination and Containment
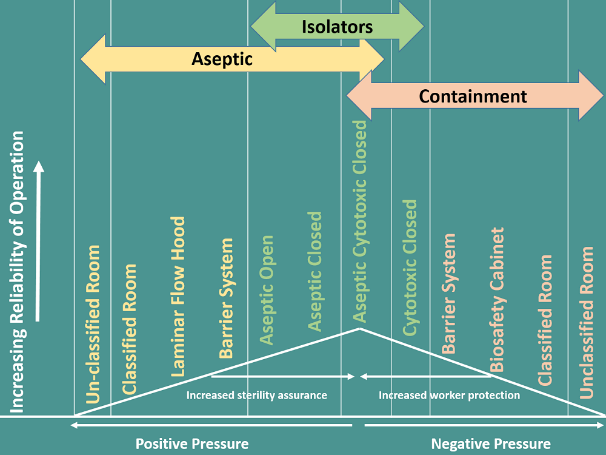
Source: Potent Product Process and Facility Design by S.C. Singhai
Esco is a world leader in Containment and Cleanroom Technologies
Esco provides containment systems for the manufacture and handling of HPAPIs. Our equipment are compliant with cGMP and other international standards, and provide comprehensive operator, product, and environment protection.
Our Products
For more information, contact us at:
Tel: +65 6542 0833
Fax: +65 6542 6920
Email: [email protected]










