The word ‘capsule’ is derived from the Latin capsula, meaning a small box. As a drug dosage form, the word refers to a formulation with an edible outer packaging made from gelatin or other suitable material. This capsule is then filled with medicine to produce a unit drug dosage form that is mainly for oral use.
Two general types of capsules:
- hard: two-piece
- soft: one-piece
Hard gel capsule filling
Hard capsule consists of two main cylindrical-shaped pieces that closes at one end.
- ‘Cap’: refers to the shorter piece that fits over the open end of the longer piece.
- ‘Body’: refers to the longer piece
The hard capsules are made in a range of fixed sizes that can be filled with a large variety of materials of different physiochemical properties.
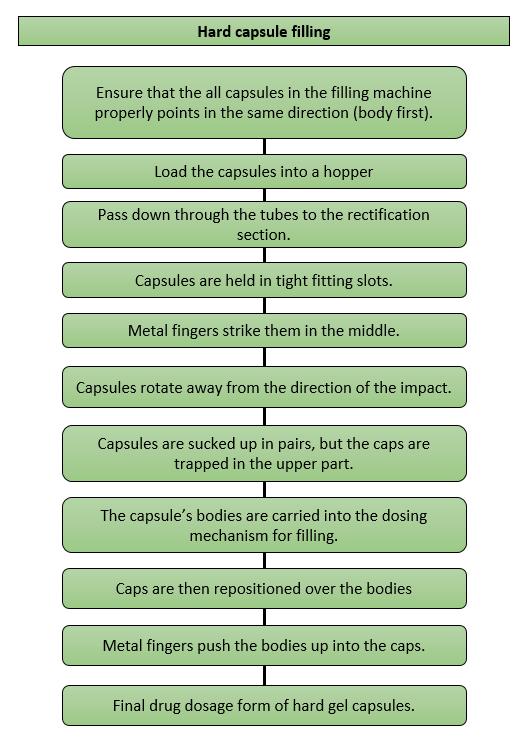
Figure 1. Hard Capsule Filling Process
Whether capsules are being filled on the bench for extemporaneous dispensing or on high-speed automatic machines for industrial products, the same set of basic operations are carried out.
There are a lot of filling methods available, the major differences between them lies on how the dose of material is measured into the capsule body.
Softgel filling
In terms of formulation requirements, the softgel can be considered as a biphasic dosage form with its solid-phase capsule shell and liquid phase capsule fill matrix.
Although it is possible to incorporate a drug in the softgel’s shell, a number of final drug products have the active ingredient(s) within the liquid-phase fill matrix. This matrix is made from a wide range of materials with different physiochemical properties.
Large-scale manufacture of liquid fill capsules is possible with the invention of the rotary die encapsulation machine. The rotary die process involves the continuous formation of a heat seal between two ribbons of gelatin concurrent with filling the softgel capsule according to the required dose. Throughout the years, even with the improvement of manufacturing processes, the basic principle for this unit operation remains.
The encapsulation process requires a certain amount of containment during filling of the hard and softgel capsules. Large amounts of concentrated powder and liquid APIs and their excipients, are utilized in the formulation, which may give rise to unwanted dust clouds or volatile substances in the atmosphere. To improve final drug product quality and prevent unwanted personnel and environmental exposure, certain primary engineering controls with proper ventilation and containment measures are critical in the manufacturing process.
References:
- Aulton, M. (2010). Aulton’s Pharmaceutics: The design and manufacture of medicines (3rd ed.). Elsevier.
Recommended Products


















