Containment Barrier Isolator (CBI) – Unidirectional
Your Practical Solution to cGMP Compliance
The Containment Barrier Isolator – Unidirectional (CBI-U) is designed to deliver a controlled ISO Class 5 (Grade A) environment through unidirectional (laminar) airflow, ensuring optimal protection for sterile and aseptic processing. By physically separating the operator from the critical process zone, the CBI-U safeguards personnel, product, and the surrounding environment within a fully enclosed barrier system. The unit can be factory-configured to operate under positive or negative pressure, with either single-pass or recirculating airflow, allowing it to be tailored to specific process requirements and containment strategies.
Engineered for small-scale aseptic operations involving non-hazardous or hazardous materials, the CBI-U provides robust containment performance and is pressure-tested to Class II according to ISO 10648-2. To further ensure chamber integrity, an optional automated pressure decay test is available for routine verification. The system can also be equipped with an integrated automated biodecontamination solution (BioVap™), achieving a validated 6-log reduction of bioburden using hydrogen peroxide, supporting rapid and reproducible decontamination cycles.
Applications:
Pharmacy Compounding (Chemotherapy/TPN)
Small-Scale Potent Material Handling
Aseptic Processing
Sterility Testing
Research and Development
Cell Processing
- Unidirectional (laminar) airflow compliant with sterile product handling standards, supported by H14 HEPA supply filters (>99.995% efficiency at 0.1–0.3 µm) delivering ISO Class 5 (Grade A) air cleanliness and double-stage H14 HEPA exhaust filtration for enhanced room protection.
- Rapid recovery time of less than 60 seconds to re-establish and maintain ISO Class 5 conditions.
- Class II containment performance in accordance with ISO 10648-2 leak tightness requirements.
- Type D pass-through chambers with dedicated H14 HEPA supply filters, featuring electromagnetic interlocking and time-delay control for safe material transfer.
- Safe multi-piece glove change system with 200 × 200 mm circular glove ports to maintain barrier integrity during replacement.
- FDA-grade static seals for both inner and outer doors to ensure reliable sealing performance.
- Integrated HMI/PLC control system with optional 21 CFR Part 11 compliance for electronic records and electronic signatures.
|
Total Exhaust/Single Pass Airflow |
Recirculating Airflow |
 |
|
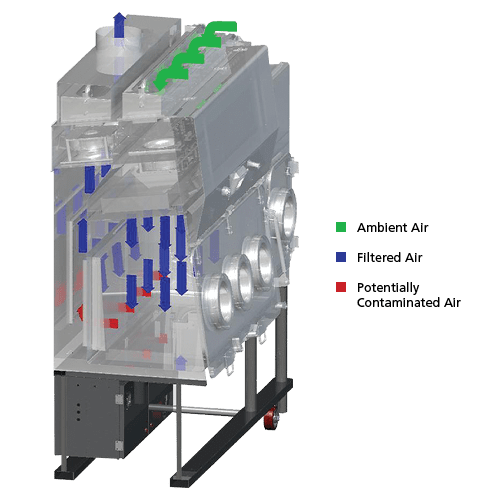
Ambient air is pulled through the inlet prefilter located on top of the isolator. Air from the top inlet and from work zone is pulled by the fan which creates a positive pressure on the plenum that creates downflow. The HEPA (H14) downflow filter creates a laminar and particle-free ISO Class 5 air cleanliness as per ISO 14644-1 inside the isolator to protect the work material inside the main chamber and pass-thru. Air from the work zone and pass-thru is quickly purged out by the fan to keep the area clean. The fan pulls approximately 70% of the purged air back to the plenum and after passing through the HEPA (H14) downflow filter.
The high rate of airflow recirculation helps to prolong filter life and reduces the chances of ambient contaminants entering the work zone. For a recirculating model, approximately 30% of the purged air is exhausted through an HEPA-filter to prevent heat build-up inside the isolator that can be detrimental to drug compounding. This exhausted air is replenished by ambient air coming from the top inlet prefilter and a filter with 80% efficiency for positive pressure model. For a Total Exhaust/Single-Pass model, 100% of the air is exhausted out of the isolator.
|
Containment Barrier Isolator - Unidirectional |
2G |
3G |
4G |
|||
|
External dimension of Process Chamber * |
1200 x 920 x 2650 mm |
1600 x 920 x 2650 mm |
2000 x 920 x 2650 mm |
|||
|
Internal dimension of Process Chamber |
1196 x 560 x 765 mm |
1596 x 560 x 765 mm |
1996 x 560 x 765 mm |
|||
|
External dimension of 1-glove Pass Chamber |
610 x 780 x 1325 mm |
|||||
|
Internal dimension of Pass chamber |
606 x 450 x 700 mm |
|||||
|
Glove port height ** |
1135 mm |
|||||
|
Glove port dimension |
Circular (200 x 200 mm) |
|||||
|
Chamber Environment |
ISO Class 5 in Process Chamber (Grade A) |
|||||
|
Chamber Pressure |
Factory-Configured Either Positive or Negative Pressure |
|||||
|
Airflow Type |
Unidirectional/ Laminar Airflow |
|||||
|
Filter Type - Chamber Inlet |
HEPA (H14) Filter with Integral Mesh Guard and Gasket Seal |
|||||
|
Filter Efficiency - Chamber Inlet |
> 99.995% at Most Penetrating Particle Size (MPPS) as per EN
1822:2009 |
|||||
|
HMI Type |
HMI Siemens/ Allen Bradley 7" |
|||||
|
Control System |
Industrial Grade PLC Siemens/ Allen Bradley |
|||||
|
Lighting Level |
≥ 500 Lux (6000 K) |
|||||
|
Sound Level |
≤ 80 dBA |
|||||
|
Isolator Construction |
Internal Chamber |
SS316L |
||||
|
Service Housings |
SS304 |
|||||
|
Support Frame |
SS304 |
|||||
|
Control Panel |
In-house SS304 (IP-20) |
|||||
|
Chamber Door |
10 mm (0.39") Tempered Glass |
|||||
|
Isolator Surface Finish |
Internal Chamber |
≤ 0.4 Ra |
||||
|
External Chamber |
≤ 0.6 Ra |
|||||
|
Electrical Requirement |
220-240 VAC, 50/60 Hz, 1Ø or 110-120 VAC, 50/60 Hz, 1Ø |
|||||
|
Compressed Air Requirement |
Min 6 Bar-g, max 12 Bar-g with 200 Liter per Minute Flow |
|||||
|
Exhaust Duct Requirements (by Client) |
250 mm (10") Duct from Isolator to Outside |
|||||
|
Estimated Weight |
850 kg |
1000 kg |
1200 kg |
|||
|
Shipping Dimension (W x D x H) |
Contact Esco for more information |
|||||
* Including exhaust collar for ducting and fixed stand with caster wheels and leveling feet
** With fixed stand with caster wheels and leveling feet
|
Building Exhaust Requirement |
2G |
3G |
4G |
|
Total exhaust (Single Pass) with 1PTC |
750 cmh |
1100 cmh |
1500 cmh |
|
Recirculating |
150 cmh |
220 cmh |
300 cmh |
Note:The values stated above are based on standard building exhaust requirements with a ±30% tolerance and may vary for customized dimensions or specific airflow configurations. Please contact Esco to confirm the exact building exhaust requirements for the proposed or selected configuration.
The Esco Containment Barrier Isolator – Unidirectional (CBI-U) is a versatile system designed to accommodate a wide range of applications, supported by various optional features and customization configurations as outlined below. Contact Esco to learn how the CBI-U can be tailored to meet your specific process and containment requirements.
|
List of Options |
CBI-U |
CBI-T |
CBI-III |
|
|
4” Split Butterfly Valve (for powder discharge of powder below
isolator chamber) |
✓ |
✓ |
✓ |
|
|
Raised and Lower Height Adjustable Support Stand |
✓ |
|||
|
Anti-blow Back Damper Box |
✓ |
✓ |
✓ |
|
|
Automated Pressure Hold Test |
With Client-supplied Compressed Air |
✓ |
✓ |
✓ |
|
With Mobile Air Compressor (Esco-supplied) |
✓ |
✓ |
✓ |
|
|
Bag Welder with Table Bag-out Port |
✓ |
✓ |
✓ |
|
|
Mobile Biodecontamination BioVap |
✓ |
✓ |
||
|
Integrated Biodecontamination BioVap |
✓ |
✓ |
||
|
Carbon Filter |
✓ |
✓ |
||
|
Integration of Small Scale Aseptic or Potent Tablet/Capsule |
✓ |
✓ |
✓ |
|
|
Double-sided Access |
✓ |
|||
|
Manual Drain Valve |
✓ |
✓ |
✓ |
|
|
Electrical Outlet |
IP-66 |
✓ |
✓ |
✓ |
|
IP-54 |
✓ |
✓ |
✓ |
|
|
Non-IP rated |
✓ |
✓ |
✓ |
|
|
ATEX Zone 21 (only for internal chamber) |
✓ |
✓ |
✓ |
|
|
ATEX rating up to zone 1/21 internally only |
✓ |
✓ |
✓ |
|
|
Glove Leak Tester |
Integrated |
✓ |
✓ |
✓ |
|
Wireless* |
✓ |
✓ |
✓ |
|
|
H2O2 Monitoring System (integrated to HMI/PLC) |
✓ |
✓ |
||
|
Entry / Exit Ports |
Liquid/ Water Waste |
✓ |
✓ |
|
|
Product Waste |
✓ |
✓ |
✓ |
|
|
N2 Purge for Process Chamber |
✓ |
|||
|
Product Waste Bag Out Ports |
✓ |
✓ |
✓ |
|
|
Particle Counter Integration |
✓ |
✓ |
||
|
Air Sampler Integration |
✓ |
✓ |
||
|
RTPØ 105, 190, 270 - Alpha |
✓ |
✓ |
✓ |
|
|
RTPØ 105, 190, 270 - Beta Canister |
✓ |
✓ |
✓ |
|
|
RTPØ 105, 190, 270 - Beta Liner |
✓ |
✓ |
✓ |
|
|
RTPØ 350 - Alpha, Beta Liner, Beta Canister |
✓ |
✓ |
✓ |
|
|
Spray Ball (CIP) with Manual Ball Valve |
✓ |
|||
|
Spray Gun (WIP) with Manual Ball Valve |
✓ |
|||
|
Sterility Test Pump |
Tabletop |
✓ |
✓ |
|
|
Integrated |
✓ |
✓ |
||
|
Temperature and Relative Humidity Monitoring (only for process
chamber) |
✓ |
✓ |
✓ |
|
|
Rear View Monitor |
✓ |
✓ |
✓ |
|
|
UV Lamp |
✓ |
✓ |
||
|
Weighing Scale Granite Slab with SS316L Frame and Leveling Feet |
✓ |
✓ |
✓ |
|
.webp)






 Thailand_1.webp)






